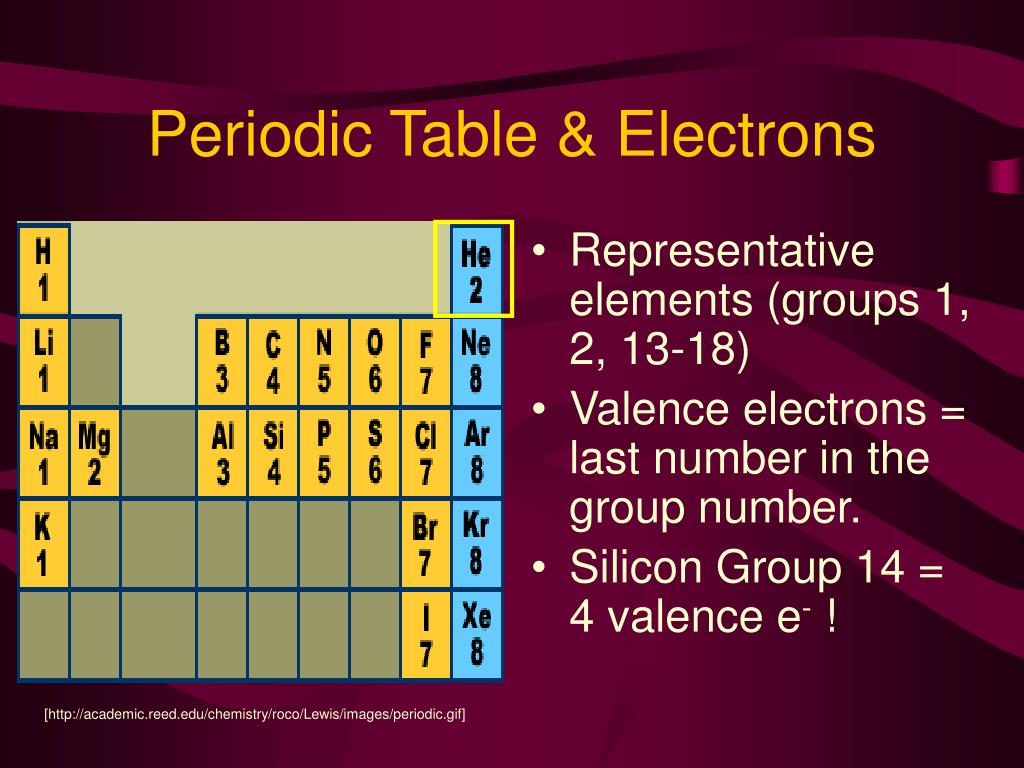
It is important to recognize which numbering system is being used and to be able to find the number of valence electrons in the main block elements regardless of which numbering system is being used. You will come across periodic tables with both numbering systems. In this numbering system, group 1A is group 1 group 2A is group 2 the halogens (7A) are group 17 and the noble gases (8A) are group 18. The elements in this group are also gases at room temperature.Īn alternate numbering system numbers all of the \(s\), \(p\), and \(d\) block elements from 1-18. We will learn the reason for this later, when we discuss how compounds form. These elements also have similar properties to each other, the most significant property being that they are extremely unreactive, rarely forming compounds. This group contains very reactive nonmetal elements.
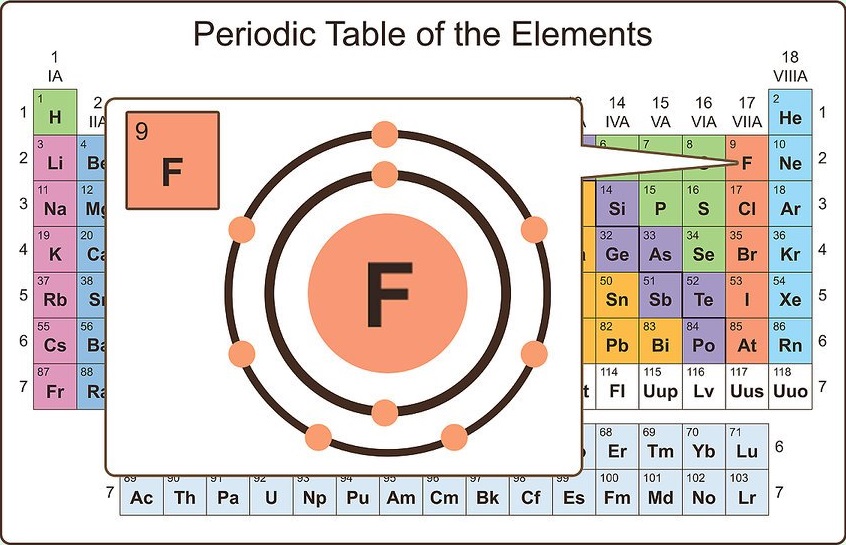
Group 7A (or 17) elements are also called halogens.
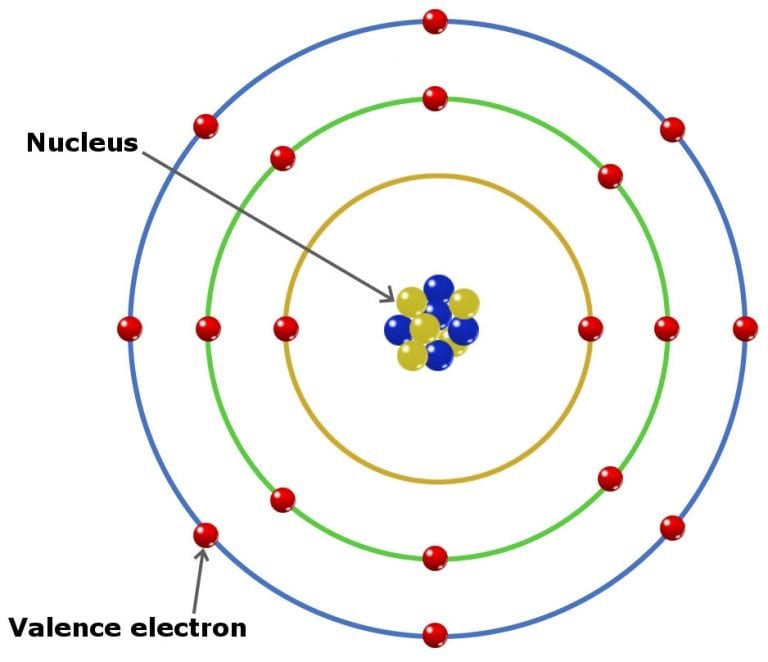
It is important to recognize a couple of other important groups on the periodic table by their group name. 9.8: The Explanatory Power of the Quantum-Mechanical Model The chemical properties of elements are determined primarily by the number and distribution of valence electrons. Electron configurations can be predicted by the position of an atom on the periodic table. Remember, Mendeleev arranged the table so that elements with the most similar properties were in the same group on the periodic table. The arrangement of electrons in atoms is responsible for the shape of the periodic table. The same pattern is true of other groups on the periodic table. Once again, because of their similarities in electron configurations, these elements have similar properties to each other. Group 2A is also called the alkaline earth metals. Although most metals tend to be very hard, these metals are actually soft and can be easily cut. Group 1A is also known as the alkali metals. Because of their similarities in their chemical properties, Mendeleev put these elements into the same group. The elements in 1A are all very reactive and form compounds in the same ratios with similar properties with other elements. This is what causes these elements to react in the same ways as the other members of the family. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group.
